FDA Recalls Over Half a Million Blood-Pressure Pills After Detecting Cancer-Linked Contaminant
The recall affects Prazosin capsules in multiple strengths, but not all generic versions on the market. Patients have been advised to check their prescription bottles for manufacturer details—Teva’s products typically carry the identifiers “MFG Teva,” “MFR Teva,” or National Drug Codes beginning with 0093.
- By Newsdesk
- New Delhi
- Dec 02, 2025
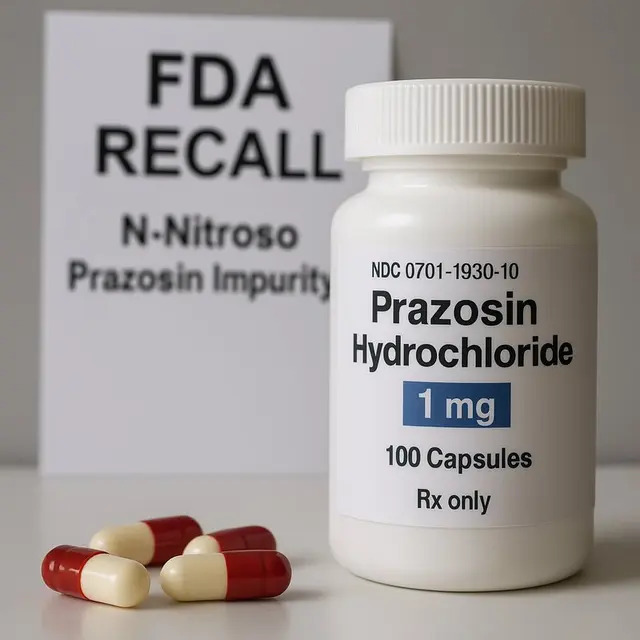
The U.S. Food and Drug Administration has announced a nationwide recall of more than 580,000 capsules of the widely prescribed blood-pressure drug Prazosin after routine testing revealed the presence of elevated levels of nitrosamines, chemicals associated with a potential cancer risk. The affected batches were manufactured by Teva Pharmaceuticals, one of the largest makers of generic medicines in the country.
Prazosin, a decades-old generic drug used by an estimated 510,000 patients each year, is primarily prescribed to treat hypertension but is also commonly used to manage symptoms of post-traumatic stress disorder. Teva reported the contamination on October 7, 2025, prompting the FDA to initiate a Class II recall, a category reserved for situations where exposure may cause temporary or medically reversible health effects. Despite the recall, regulators emphasised that patients should not stop taking the medication abruptly, as untreated blood-pressure spikes can be far more dangerous.
Nitrosamines, the contaminants detected in the capsules, can form when certain chemical components interact during the manufacturing process or storage. Although these chemicals are also found in small amounts in everyday items such as preserved foods, sustained exposure to higher concentrations has been linked to increased cancer risk. Similar issues surfaced in 2018 when several blood-pressure drugs were found to contain nitrosamine impurities, prompting the FDA to tighten testing guidelines and issue updated safety directives in 2024.
The recall affects Prazosin capsules in multiple strengths, but not all generic versions on the market. Patients have been advised to check their prescription bottles for manufacturer details—Teva’s products typically carry the identifiers “MFG Teva,” “MFR Teva,” or National Drug Codes beginning with 0093. Those with affected batches are encouraged to contact their pharmacists or physicians to discuss replacement options, which may include unaffected formulations or alternative medications such as Clonidine or Trazodone.
The incident has reignited concerns about the safety of older generic drugs, many of which have shifted between manufacturers over the years and are now produced in facilities across several countries. A recent study highlighted that generics manufactured in India were associated with a significantly higher rate of serious adverse events compared with those made in the United States, raising questions about quality disparities linked to cost-driven production models. Experts argue that intense price competition in the generic market may be pushing manufacturers toward practices that compromise safety, particularly for long-established drugs with thin profit margins.
Because pharmacy labels often do not disclose where a drug was manufactured, consumers and even pharmacists may have little insight into the quality differences between versions of the same medication. The Prazosin recall, researchers warn, underscores the need for greater transparency and more robust oversight to ensure that legacy generics remain safe for long-term use.
As regulators investigate the root cause of the contamination, patient safety advocates stress that the recall should serve as a broader warning: longstanding generic medicines, often assumed to be low-risk, may carry hidden vulnerabilities that only emerge after decades of complex supply chain changes. For now, patients are urged to review their medication labels and consult their healthcare providers before making any changes to their treatment plans.
